 |
 |
- Search
| J Neurosonol Neuroimag > Volume 14(2); 2022 > Article |
|
Abstract
The clinical manifestations of cerebral lupus vasculitis have a wide spectrum of severity which can result in different prognoses. Therefore, its diagnosis and treatment remain challenging without specific guidelines. A 49-year-old woman with systemic lupus erythematosus presented with transient left side paresthesia and dysarthria lasting 20 minutes. Brain magnetic resonance imaging (MRI) showed a multifocal diffusion restriction and sulcal hyperintensities in the right middle cerebral artery region. Brain magnetic resonance angiography (MRA) showed multifocal steno-occlusive lesions at her intracranial arteries. Because she only showed transient symptoms, aspirin (100 mg/day), atorvastatin (20 mg/day) and an increased dose of prednisolone (5 mg/day to 10 mg/day) were started, on the suspicion of cerebral lupus vasculitis. Brain MRIs/MRAs renormalized after four months without any recurrence. Cerebral lupus vasculitis manifests diverse clinical and neuroimaging characteristics. Although brain image demonstrates devastating features, tailored immune therapy should be selected based on the severity of the patient’s symptoms.
Systemic lupus erythematosus (SLE) is a systemic autoimmune disease caused by autoantibody production which leads to an attack on any organ of the body. Although central nervous system (CNS) involvement is common and results in various neuropsychiatric symptoms in SLE patients,1 cerebral vasculitis is rare and its incidence in postmortem studies is known to be less than 10%.2 Since cerebral vasculitis predominantly involves small vessels rather than large cerebral arteries, diagnosis with conventional angiography, which detects segmental stenosis and dilation of multiple vascular regions, is often unable to detect cerebral vasculitis.3 However, recent advanced magnetic resonance imaging (MRI) techniques including vessel wall MRI are simple to implement and may provide additional information on the vascular changes of small vessels, such as concentric wall thickness and wall enhancement.4 Therefore, the prevalence of cerebral vasculitis in SLE may actually be significantly higher than has been previously reported. Herein, we report a case of cerebral infarction caused by cerebral vasculitis which was confirmed by magnetic resonance angiography (MRA) with sulcal hyperintensities in a fluid attenuated inversion recovery (FLAIR) image.
A 49-year-old woman presented to the emergency department with transient left side paresthesia and dysarthria lasting 20 minutes. She had been diagnosed with lupus nephritis 12 years prior, for which she had been undergoing treatment with prednisolone (5 mg/day) and hydroxychloroquine (300 mg/day). On physical examination, her temperature was 36.3°C, heart rate was 74/min, and blood pressure was 217/122 mmHg. Laboratory findings revealed a decreased white blood cell count (3,220/mm3), normal hemoglobin (12.7 g/dL), normal platelet count (158,000/mm3), normal erythrocyte sedimentation rate (8 mm/hour) and normal C-reactive protein (0.2 mg/dL). Other laboratory findings such as lipid profile, electrolyte, liver function, renal function, and coagulation profiles were unremarkable. Immunologic laboratory findings were negative for antiphospholipid antibodies, anti-dsDNA antibody, anti-SSB/La antibody, anti-RNP antibody, anti-PR3 antibody, and anti-MPO antibody. However, anti-nuclear antibody, anti-SSA/Ro antibody, and anti-Sm antibody tests were positive with low complement levels (C3, 53 mg/dL; C4, 7 mg/dL).
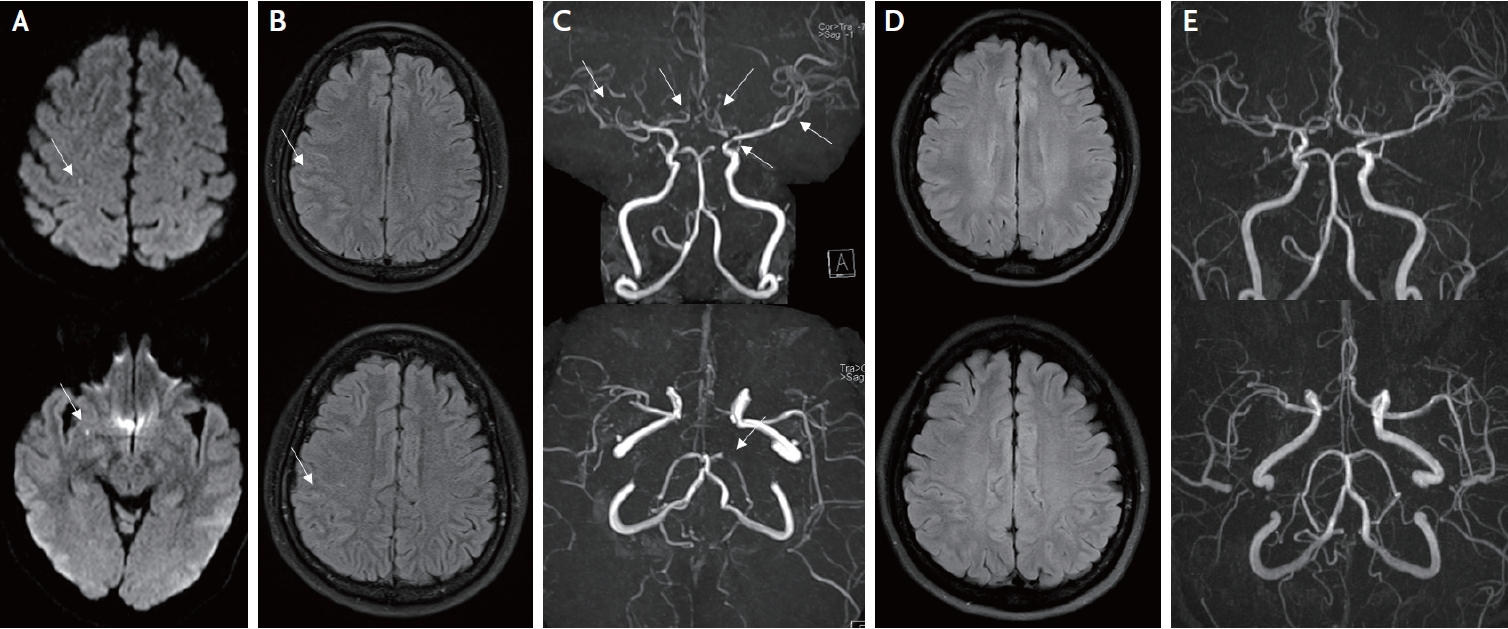
Brain diffusion-weighted imaging showed focal dot-like restrictions in the right temporal and parietal lobes (Fig. 1A). FLAIR imaging showed sulcal hyperintensities in the right middle cerebral artery region (Fig. 1B). Brain MRA revealed multifocal stenosis on both anterior cerebral arteries, the middle-right cerebral artery, the distal-left internal carotid artery and the left posterior cerebral artery (Fig. 1C). The patient was diagnosed with lupus vasculitis rather than reversible cerebral vasoconstriction syndrome because she had no headache, no predisposing factors, and had been treated for SLE for some time. Although her neuroimaging findings showed severe disease progression involving large arteries, she only had minimal outward manifestations without any neurological symptoms. Because of this, she was treated only with an antiplatelet agent (aspirin 100 mg/day), statin (atorvastatin 20 mg/day) and increased dose of prednisolone (10 mg/day).
After four months, a follow-up brain MRI showed improvement of the patient’s sulcal hyperintensies and multiple steno-occlusive lesions. FLAIR imaging showed no abnormal findings (Fig. 1D), and MRA showed interval improvement of her cerebral arteries (Fig. 1E). Oral prednisolone was reduced to 5 mg/day and stroke symptoms did not recur during the next 18 months of follow up.
This case involved, a middle-aged woman patient with SLE who presented with cerebral infarction associated with cerebral vasculitis with sulcal hyperintensities, which was improved with a low-dose steroidal treatment (prednisolone 10 mg/day). Her neurological symptoms subsided completely without any recurrence for 18 months after initial stroke symptoms.
Direct disease-associated primary neuropsychiatric SLE (NPSLE) is generally classified as either an immune or inflammation-mediated disease (inflammatory NPSLE) or a thrombotic cerebrovascular disease (thrombotic NPSLE).1 However, cerebral vasculitis, ambiguously, can manifest both inflammatory and thrombotic features with complex and heterogenous findings.5 It can manifest lupus symptoms through inflammatory processes triggered by immune complex deposition in the blood vessels,6 or develop atherosclerotic vascular disease in a proinflammatory environment.7 Thus, cerebral vasculitis may have a low rate of confirmation as a cause of CNS vascular injury. In addition, cerebral vasculitis is known to impair small vessels rather than large arteries.8 This may also result in a low diagnostic rate because conventional angiography is often limited in its ability to image small arteries.3 Recent advance to brain MRI technology may reveal cerebral vasculitis using various novel techniques. MRI and MRA with or without contrast enhancement can be widely and noninvasively applied to patients, and may detect parenchymal abnormalities and vascular abnormalities such as beaded appearance with variable degrees of stenosis and occlusion.9 In our patient, multiple large artery stenotic lesions and multiple small diffusion restriction and sulcal hyperintensities were detected, which is consistent with large artery disease. Since advanced imaging is now used widely, detecting large artery vasculitis may become easier than it has been in the past.3 High resolution MRI with vessel wall imaging is another emerging diagnostic tool which directly visualizes vessel walls and allows for discrimination between different diseases such as cerebral vasculitis, intracranial atherosclerosis, intracranial dissection, moyamoya disease, and reversible cerebral vasoconstriction syndrome. In addition, intracranial vessel wall MRIs can also detect cerebral vasculitis in small vessels by wall enhancement, which cannot be visualized by conventional or magnetic resonance angiography.10 Although we did not use high resolution MRI in this patient, the use of this recently developed diagnostic method may increase the rate of diagnosis of true vasculitis.
The treatment of cerebral vasculitis is also challenging because there are no specific guidelines. Its wide spectrum of clinical manifestations and involvement of different vessel sizes may be significant to treatment direction.5 Based on the fact that cerebral vasculitis can present both the inflammatory and thrombotic features of SLE, a combination of immunosuppression and anti-thrombotics may be needed in this patient group.1 Although high doses of glucocorticoids and monthly intravenous cyclophosphamide for six months are recommended for inflammatory NPSLE including cerebral vasculitis,1 we believe that the severity of the symptoms should be considered before determining treatment methods. Our patient serves as an example where treatment with low-dose prednisolone (5 mg/day), an antiplatelet agent, and statin completely reversed large vessel steno-occlusive lesions after four months, without any recurrent symptoms.
Proper diagnosis and treatment of cerebral vasculitis is crucial for every lupus cerebral vasculitis attack. Newly developed advanced imaging techniques may improve diagnostic accuracy and appropriate treatment for this condition.
NOTES
Ethics statement
This case was approved by the Institutional Review Board of Hanyang University Hospital (IRB No. HYUH- 2022-10-017). The need for informed consent was waived by the board.
Fig. 1.
Brain magnetic resonance imaging and magnetic resonance angiography (MRA) of the patient. Initial diffusion-weighted image shows multiple dot-like high signal intensity lesions in the middle-right cerebral artery region (A, arrows). Initial fluid attenuated inversion recovery (FLAIR) imaging reveals sulcal hyperintensities in the middle-right cerebral artery region (B, arrows). Initial time-offlight MRA shows multiple steno-occlusive lesions at both anterior cerebral arteries, both middle cerebral arteries, left distal internal carotid artery, and left posterior cerebral artery (C, arrows). Follow up FLAIR image shows normal appearance without sulcal hyperintensities (D). Follow-up MRA also shows significant improvement of steno-occlusive lesions in intracranial arteries (E).
REFERENCES
1. Schwartz N, Stock AD, Putterman C. Neuropsychiatric lupus: new mechanistic insights and future treatment directions. Nat Rev Rheumatol. 2019;15:137-152.




2. Rowshani AT, Remans P, Rozemuller A, Tak PP. Cerebral vasculitis as a primary manifestation of systemic lupus erythematosus. Ann Rheum Dis. 2005;64:784-786.



3. Edgell RC, Sarhan AE, Soomro J, Einertson C, Kemp J, Shirani P, et al. The role of catheter angiography in the diagnosis of central nervous system vasculitis. Interv Neurol. 2016;5:194-208.




4. Arslan S, Korkmazer B, Kizilkilic O. Intracranial vessel wall imaging. Curr Opin Rheumatol. 2021;33:41-48.


5. Leone P, Prete M, Malerba E, Bray A, Susca N, Ingravallo G, et al. Lupus vasculitis: an overview. Biomedicines. 2021;9:1626.



7. Pyrpasopoulou A, Chatzimichailidou S, Aslanidis S. Vascular disease in systemic lupus erythematosus. Autoimmune Dis. 2012;2012:876456.




8. Böckle BC, Jara D, Aichhorn K, Junker D, Berger T, Ratzinger G, et al. Cerebral large vessel vasculitis in systemic lupus erythematosus. Lupus. 2014;23:1417-1421.










