Transcranial Doppler Ultrasound Role for Patients with Traumatic Brain Injury
Article information
Abstract
Neurologists, neurosurgeons, and neurointensivists, including military, have a large armamentarium of diagnostic and monitoring devices available to detect primary and secondary brain injury and guide therapy in patients with acute traumatic brain injury (TBI) to avoid cerebral ischemia due to the posttraumatic vasospasm (PTV) and intracranial hypertension (ICH). This review summarizes the advantages and the specific roles of transcranial Doppler (TCD) ultrasonography for patients with acute and longterm effects of TBI. In critical care setting numerous publications showed that TCD is predictive of angiographic PTV and onset of ICH. The post TBI status of cerebrovascular reactivity and cerebral hemodynamics also has important implications with regard to the treatment of long-term effects of mild TBI (mTBI). Today it is abundant evidence that TCD is an important tool for monitoring the natural course of acute moderate and severe TBI, for evaluating the effect of medical treatment or intervention, for forecasting, and for identifying high-risk patients for onset of cerebral ischemia after TBI. TCD makes good clinical and economic sense as it is a reliable, quantitative, non-invasive and non-expensive “biomarker” to the acute clinical manifestations of TBI. TCD clinical utilization holds promise for better detection, characterization, and monitoring of objective cerebral hemodynamics changes in symptomatic patients with TBI not readily apparent by standard CT or conventional MRI techniques. TCD utilization will improve the sensitivity of neuroimaging to subtle brain perturbations and combining these objective measures with careful clinical characterization of patients may facilitate better understanding of the neural bases and treatment of the signs and symptoms of TBI. This review summarizes the advantages and the specific roles of TCD ultrasonography for patients with acute and long-term effects of TBI.
TBI EPIDEMIOLOGY
Traumatic brain injury (TBI) is a major cause of death and disability in the United States. There were approximately 223,135 TBI-related hospitalizations in 2019 and 64,362 TBI-related deaths in 2020.1 This represents more than 611 TBI-related hospitalizations and 176 TBI-related deaths per day. These estimates do not include the many TBIs that are only treated in the emergency department (ED), primary care, urgent care, or those that go untreated.2 In 2014, about 2.87 million TBI-related ED visits, hospitalizations, and deaths occurred in the USA, including over 837,000 of these health events among children.3 TBI was diagnosed in approximately 288,000 hospitalizations, including over 23,000 among children. TBI is a contributing factor in 30.5% of injury-related deaths among civilians.4 Additionally, since 2000, over 260,000 US military service members were diagnosed with TBI, with the vast majority classified as mild or concussive (76%).5 In Europe TBI is also an important cause of death and hospital admissions. In 2012, 1,375,974 hospital discharges (data from 24 countries) and 33,415 deaths (25 countries) related to TBI were identified.6 Estimate 56,946 TBI-related deaths and 1,445,526 hospital discharges occurred in 2012 in the European Union (population 508.5 million) and about 82,000 deaths and about 2.1 million hospital discharges in the whole of Europe (population 737 million).6 Interesting fact that in South Korea the age-adjusted incidence per 100,000 people increased until 2010 but later showed a decreasing trend (475.8 cases in 2017) thereafter; however, a continuously decreasing age-adjusted mortality trend observed (42.9 cases in 2008, 11.3 in 2017).7 The authors observed changing trends in the TBI incidence, with a continuously decreasing overall incidence and a rapidly increasing incidence and high mortality values in older adults.7 Their findings highlight the importance of active TBI prevention in elderly people.
Those who survive a TBI can face effects that last a few days, or the rest of their lives. Effects of TBI can include impairments related to thinking or memory, movement, sensation (e.g., vision or hearing), or emotional functioning (e.g., personality changes, depression). These issues not only affect individuals but also can have lasting effects on families and communities. Sixty-nine million individuals are estimated to suffer TBI from all causes each year, consequently it is clear that TBI represent the “silent epidemic” that contributes to worldwide death and disability more than any other traumatic insult.8 Therefore management of moderate and severe TBI in the field, at the site of motor-vehicle accident, during transportation, in the ED, and in the first 48 hours of critical care can have significant consequences on outcomes, including mortality, complications, and long-term recovery and function.
TBI PATHOPHYSIOLOGY
Outcome from TBI is determined by two substantially different factors: (1) the primary insult occurring at the moment of impact and (2) the secondary insult represents consecutive pathologic processes initiated at the moment of injury with delayed clinical presentation. A single center prospective observational study in patients with moderate to severe TBI showed that 70% of patients experienced neurological complications and outcome was worse on those patients.9 For moderate-to-severe TBI patients delayed cerebral ischemia (DCI) due to the presence of posttraumatic vasospasm (PTV) and intracranial hypertension (ICH) are major contributing factors for secondary injury. In addition, inflammation,10 parenchymal contusions and fever are defined as independent risk factors for development of PTV.11 In patients with acute brain injury, altered cerebral spinal fluid concentrations of protein biomarkers related to cytoskeletal damage, inflammation, apoptosis and oxidative stress may be predictive of worse neurological outcomes.12 Other authors are indicating that mechanical stretching, calcium dysregulation, endothelin, contractile proteins, products of cerebral metabolism and cortical spreading depolarization have been involved in PTV pathophysiology.13 Therefore, PTV could be evident even without of presence posttraumatic SAH (tSAH). To conclude, the exact reason for the occurrence of DCI is still not clearly understood, and several theories exist.14-19
TBI NEUROIMAGING
While history, mechanism and physical exam remain mainstays of diagnosis in TBI, medical imaging remains a key modality for assessment of TBI in both the clinical and research settings. Currently, there is no definitive objective diagnostic tool for TBI that effectively describes the multiple domain insults that a TBI casualty can suffer, to include polytrauma and effects of emergent field management and transport. Cranial computed tomography (CT) scanning is the most common imaging modality used during the acute phase of TBI to detect subcutaneous or subgaleal hemorrhage, skull fractures, epidural and subdural hemorrhage, and parenchymal injury.20-23 There is high-quality evidence that CT scanning is clinically valuable in the evaluation of patients presenting to ED with moderate and severe TBI.23,24 In these patients, CT is highly sensitive in identifying intracranial hemorrhages that require neurosurgical interventions, and can often be life-saving.25 High-quality evidence is also present in the literature that CT scanning is not useful for the prognosis prediction or surgical intervention of TBI patients presenting to the ED.26 CT scanning is of limited usefulness in the clinical evaluation of patients presenting to the ED with mTBI and cranial CT scanning is likely overused in the evaluation of mTBI.27 Given the inherent limitations of CT scanning, it is possible that further studies designed to identify patients with mTBI who are at risk of persistent post-concussive symptoms and long-term disabilities will be in the area of blood biomarkers, which already show some promise for detecting patients with mTBI who can be safely discharged without a CT scan.28,29
Magnetic resonance imaging (MRI) offers moderate clinical utility in both CT-negative and CT-positive TBI.30,31 However, MRI shows significant utility for research into the evaluation of TBI. Research arms should include clinical correlation with neuropsychological symptoms and outcomes in longitudinal studies of patients with (or at risk for) mTBI.32 In addition, validation of MRI observations correlated with pathology are essential both to confirm and to understand the cellular processes of astrocytic and neuronal injury in TBI.32 Occasionally, edema-sensitive sequences, such as Diffusion-Weighted Imaging (DWI) and Fluid Attenuated Inversion Recovery (FLAIR), or blood-sensitive sequences, such as Gradient Echo (GRE) and Susceptibility Weighted Imaging (SWI), will discover small cortical contusions that are obscured by the adjacent bone on a CT scan, or small white-matter lesions in characteristic locations for diffusion axonal injury (DAI). This ability to view potentially obscured contusions is an important advantage of brain MRI: only 10% of DAI is positive on CT because more than 80% of lesions are non-hemorrhagic and are therefore better detected with a combination of DWI, FLAIR, and GRE.33 However, other authors concluded that there is no association of DAI and long-term TBI outcomes, and therefore physicians should be cautious in attributing DAI to survival, future neurologic function, and quality of life.34
Positron Emission Tomography (PET) has been used for over three decades as a research tool to study the pathophysiology of an array of brain diseases, including TBI.35-37 It has led to a deeper understanding of the physiological and biochemical abnormalities underlying these conditions. PET remains a powerful tool for clinical research in TBI. PET measurements of CBF, oxygen metabolism, and glucose metabolism have been used to study the pathophysiology and effect of treatment in acute TBI, and the relationship between clinical deficits and brain abnormalities in chronic TBI.37-39 However, clinical utility of PET in the management of individual patients with TBI, however, has not yet been demonstrated.19,21,40
Another neuroimaging modality is a Singly Photon Emission Computed Tomographt (SPECT) has been employed as a diagnostic method to aid in the clinical diagnosis of TBI and as a research tool.41-44 SPECT may reveal functional abnormalities with or without the presence of anatomical abnormalities identified on conventional morphological neuroimaging techniques. However, further studies needed using a systematic approach of interpreting brain SPECT imaging based on clinical presentation of patients with TBI.
TBI subpopulation heterogeneity related to force type, severity, injury location, clinical course, clinical comorbidities, and prognosis represent distinct challenges in determining the extent of injury in a particular individual. Validated neuroimaging or other biomarkers are still needed for diagnosing of mTBI and characterization of neural network injury for managing (including monitoring and response to treatment and prognosis) all severities of TBI. Lack of these validated neuroimaging and biomarkers confounds the progress of research, including developing new treatments, but the field is quickly evolving. However, the objective assessment of TBI via imaging or other modalities is still a critical research gap, both in the civilian and military communities. In 2011, the USA Department of Defense (DoD) prepared a congressional report summarizing the effectiveness of seven modalities (CT, MRI, transcranial Doppler [TCD], PET, SPECT, electrophysiological techniques and functional near-infrared spectroscopy) to assess the spectrum of TBI from concussion to coma.45 In this report, neuroimaging experts identified the most relevant peer-reviewed publications and assessed the quality of the literature for each imaging technique in the clinical and research settings. Although CT and TCD were determined have most useful in clinical setting for moderate and severe TBI, no single imaging modality proved sufficient for all patients due to the heterogeneity of TBI. Based on the published data included in the review, MRI was only of moderate clinical utility, but only CT and TCD were of high clinical utility.45 Paper by Amyot and co-authors published in 2015 also confirmed results describes in DoD Report to US Congress.32 Our review expands on the clinical and research utility of TCD technique for TBI patients.
TCD AND POSTTRAUMATIC VASOSPASM
It was well known that cerebral ischemia due to the onset of PTV and ICH are major contributing factor for secondary injury and onset of DCI.46-52 The extent and timing of posttraumatic cerebral hemodynamic disturbances have significant implications for the monitoring and treatment of patients with TBI. Amongst TBI patients, tSAH is one of the commonest traumatic brain lesions.50,53-57 T-SAH frequently occurs in TBI patients and varies between 39% to 65%.32,33 Subsequently rates of PTV ranging from 27% to 63%,50 with a rate of 36.3% in the pediatric population.58 TBI patients with tSAH have worse outcomes compared to those without this lesion.53-59 Merely 15% of patients with tSAH achieved good outcome.56 Cerebral PTV and subsequent DCI have been shown to be leading factors leading to worst patient outcomes, which occur in up to 40% of severe TBI patients.59 In addition, PTV occurred in a substantial number of patients with war-time neurotrauma, and clinical outcomes were worse for those with this condition.59,60 These findings demonstrate that cerebral arterial vasospasm (VSP) is a frequent and significant complication of combat TBI; therefore, daily TCD monitoring is recommended for their recognition and subsequent management (Fig. 1). The degree of PTV after tSAH similar to degree of VSP after aneurysmal SAH (aSAH) is graded into mild, moderate and severe based on combination of two TCD parameters including mean cerebral blood flow velocity (CBFV) (cm/sec) and Lindegaard ratio (Table 1).61 The occurrence of PTV after any type of TBI is not infrequent and close TCD monitoring should be considered for the treatment of such patients. As it was determined by Oertel et al.62 the incidence of PTV after TBI is similar to that following aSAH and because PTV is a significant event in a high proportion of patients after TBI, close CBFV TCD monitoring is recommended for the treatment of such patients.59-64

Patient with right side contusion, base skull fracture, L1-L5 fracture. (A) Left vertebral artery injection showing severe vasospasm affecting basilar artery before angioplasty and corresponding transcranial Doppler (TCD) measured cerebral blood flow velocity (CBFV) with abnormal high resistant TCD waveform in the basilar artery. (B) Resolution of vasospasm after transluminal angioplasty and TCD showed CBFV normalization and normal low resistant TCD waveform after angioplasty.
Therefore, TCD is an extremely useful modality in monitoring the temporal course of PTV and ICH after TBI in adult and pediatric population.64-71 Even though repeat angiography is unavoidable in most TBI with tSAH patients, TCD can guide the timing of this procedure and the tailoring of aggressive treatment regimens.59,60,64 The key is not only to predict compromised perfusion by TCD, but to identify patients going into PTV and to quickly confirm PTV when subtle signs are present, before apparent neurologic deterioration. It is useful to perform TCD test on admission (or as soon as possible after surgery) and perform daily TCD studies when the patient is in the ICU.64 Similar to aSAH early TCD studies are recommended in TBI. It is therefore recommended, that serial TCD examinations be started in the first 72 hours post injury,65 or immediately upon admission.66-68 The frequency with which TCD should be performed may be guided by patient clinical presentation, knowledge of risk factors for PTV, and early clinical course. Infrequently, clinical PTV occurs earlier than natural history would suggest; daily TCD can be the least expensive option to identify patients at risk for deterioration. The necessity for medical or endovascular intervention in patients with moderate-to-severe TBI can be made logical when CBFVs abnormalities are identified daily. Therefore, the presence and temporal profile of CBFVs recorded by TCD in all available vessels of anterior and posterior circulation must be detected and serially monitored and its trends must be observed (Fig. 2). TCD studies should be performed after endovascular treatment to confirm success of intervention or identify patients with recurrent PTV and a biphasic CBFV temporal profile (Fig. 2). Although the second peak usually is not associated with a worsening of symptoms, these patients were more likely to exhibit clinical symptoms during the first CBFV peak.72 The high sensitivity of TCD to identify abnormally high CBFVs due to the onset of PTV demonstrates that TCD is an excellent first-line examination to identify those patients who may need urgent aggressive treatment and it could be used in ED.67-69 In addition, a dedicated and experienced team of neurologists, neurosurgeons, neurointensivists and neuroradiologists are required to provide the best available care and better outcome for those patients suffering TBI and interested in reducing the adverse outcomes associated with tSAH.
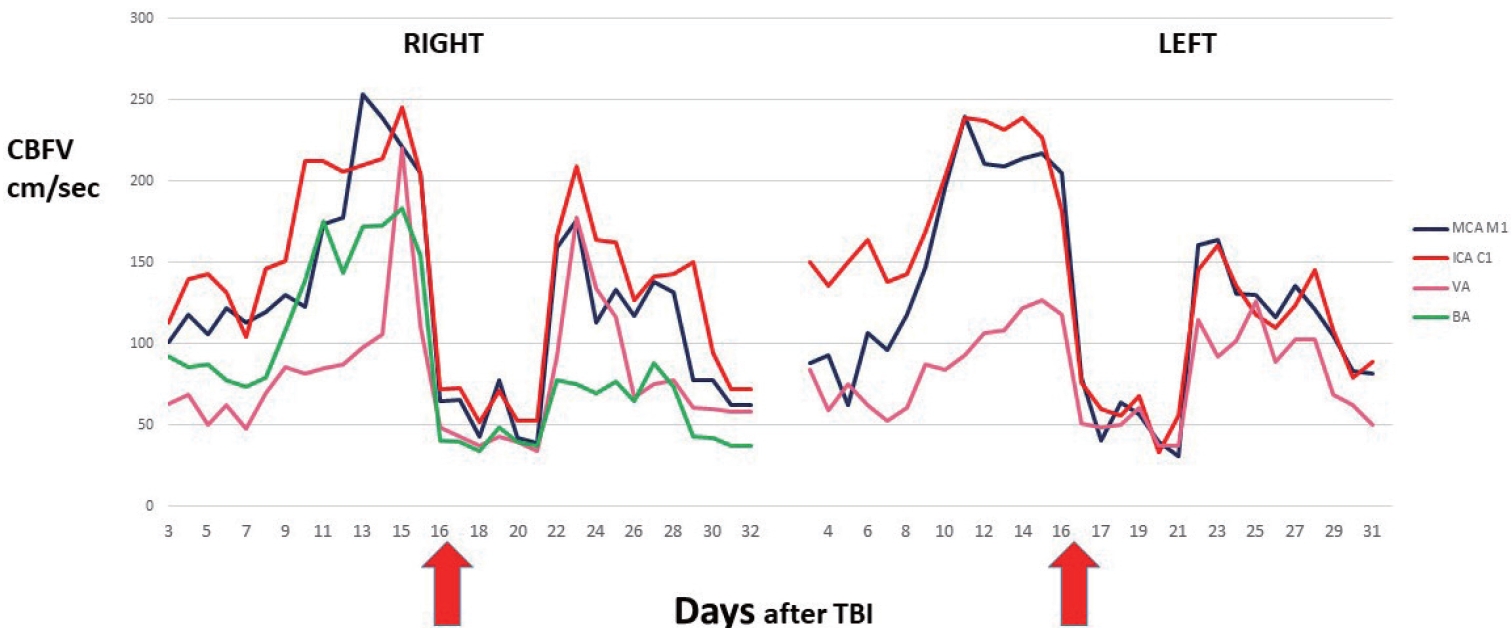
Patient with status post improvised explosive device blast injury with penetrating fragments. CT and CTA demonstrate diffuse left posterior SAH, subdural hematoma as well as multiple focal parenchymal hemorrhages within the bilateral basal ganglia, internal capsulae and right cerebellar hemisphere. On day 16 after TBI cerebral angiography and bilateral transluminal angioplasty (TLA) of anterior circulation and VB system was performed and TCD showed immediate positive effect of treatment (red arrows indicate day of TLA). On day 22 after TBI TCD demonstrate PTV recurrence. CT, computed tomography; CTA, CT angiography; SAH, subarachnoid hemorrhage; TBI, traumatic brain injury; TCD, transcranial Doppler; PTV, posttraumatic vasospasm.
Future studies are clearly needed to determine the extent to which PTV causes cerebral ischemia and infarction, and whether its development is directly affected by tSAH. However, although no adequate study has been conducted, TCD is thought to be valuable standard in the day-to-day evaluation of tSAH patients and to assess the effect of tSAH and to diagnose and monitor PTV.45,46,50,51,57-60,70-81
PTV following TBI is a very important source of morbidity and mortality. Too often, the first sign is a neurologic deficit, which may be too late to reverse. TCD assists in the clinical decision-making regarding further diagnostic evaluation and therapeutic interventions. As TCD-defined vasospasm after aSAH preceded the neurological deficit in 64%,82 earlier intervention might reduce the incidence of PTV related stroke in civilian or military hospitals with similar practice patterns. Numerous studies have shown the effectiveness of TCD in diagnosing cerebral PTV both in anterior and posterior circulation following aSAH (Quality of evidence: class II; Strength of recommendation: type B).69,83-85 Data on sensitivity, specificity, and predictive value of TCD for PTV after tSAH are needed. Data are insufficient regarding how use of TCD affects clinical outcomes after tSAH (Type U). However, up to the present time we do have overwhelming evidence based mainly on observational studies to support daily TCD testing for patient with TBI (mild, moderate and severe).18-49,52-55
TCD AND INTRACRANIAL HYPERTENSION
Raised intracranial pressure (ICP) is a life-threatening condition that can result in brainstem compression and compromised brain circulation. Increased ICP is associated with increased morbidity and is an independent predictor of mortality and of a composite endpoint of functional and neuropsychological outcome at the 6-month follow-up in moderate or severe TBI patients.86 However, even in patients initially diagnosed with mild or moderate TBI, slow growth of a hematoma with consequent development of ICH will adversely affect outcome.67,68 Monitoring of ICP is, therefore, a reasonable approach to discovering a progressive increase in ICP in TBI patients.87 It is shown that TCD as the non-invasive ultrasound modality capable of identifying patients who are progressing to ICH, and it can also monitor the effectiveness of any pharmacological intervention, and detect normalization of the ICP and Pulsatility Index (PI). The primary purpose of TCD ultrasonography is to determine the linear CBFV by quantitative interpretation of TCD waveforms. Although the qualitative contour of the TCD waveform during ICP elevation falls into a recognizable pattern, the interpretation depends on the experience and expertise of the TCD examiner and interpreter. Objective, reproducible, and verifiable measures of TCD waveform changes are necessary for TCD findings to be used with certainty for evaluation of intracranial hypertension and high ICP. One method of quantifying these changes is utilization of the PI. PI It is a calculated index of the TCD waveform.88 The pulsatility of the waveform reflects the amount of resistance in the more distal cerebral blood vessels. PI takes into account the peak systolic CBFV and the end-diastolic CBFV and compares the changes in these variables against the change in the standard measure of the entire waveform, such as mean CBFV. With the ICP higher than 15–20 mmHg, the PI has been evaluated as an alternative to direct ICP measurement.90-92 There is also a significant correlation between the cerebral perfusion pressure (CPP) and PI.91-95 In a prospective study, it was shown that TCD ultrasonography is valid in predicting the patient’s outcome of 6 months and correlates significantly with ICP and CPP values when it is performed within the first 24 hours after severe TBI.89 However, to use PI as indirect evaluation of ICP and CPP three conditions must be fulfilled: mean arterial pressure, carbon dioxide tension, and cardiac output must be within normal limits or at least not significantly different compared to the previous day. Several publications indicate the clinical value of TCD for measuring the middle cerebral artery (MCA) CBFV and PI as possible predictors of outcome in severe TBI management.70,71,75,90,95-104 Some authors suggest that early use of PI measurement permits identification of patients with low CPP and high risk of cerebral ischemia, and in emergency situations PI can be used alone, when ICP monitoring is contraindicated or not readily available.67,68,100
TCD can be used to evaluate ICP, either independent of or in conjunction with other invasive and non-invasive imaging studies.105-118 To the best of our knowledge, nobody yet suggests use of PI as an accurate method to quantitatively express ICP in mmHg. Nevertheless, in numerous publications it was shown that PI correlates well with ICP as measured by invasive methods and support use of TCD PI measurement as a predictor of elevated ICP and showed positive linear correlation between PI and ICP.62,63,66,98,119-122 At present the role of TCD for the detection of high ICP and low CPP due to the presence of ICH could be suggested as:
1) TCD waveform changes indicates abnormally high ICP, especially above 20 to 30 mmHg (PI must be 1.2 or higher and end-diastolic CBFV 25 cm/sec or lower for all anterior circulation vessels uni- or bilaterally);67,68,94
2) TCD changes may alert Neuro-ICU personnel and may indicate malfunctioning of ICP probe;105,106
3) Abnormally globally decreased pattern of the CBFV’s in parallel with increased PI’s indicates onset of diffuse ICH;107-109
4) Sudden onset of asymmetrical CBFV’s and PI’s changes may indicate a potential mid-line shift.110-112 However, we would like to stress that at present TCD alone cannot substitute invasive and quantitative ICP monitoring modalities.113 Nevertheless, in cases when invasive ICP measurements are unobtainable TCD can be used to evaluate ICP either independent of or in conjunction with other non-invasive imaging studies.113-119
The strong correlation observed between ICP and PI through the management period of patients with suspected ICH can lead us to use TCD ultrasonography-derived PI as a guide if invasive ICP monitoring is not available. At the same time, some authors argue that PI is not a reliable predictor of ICP.123,124 However, we completely agree with Kristiansson et al.’s117 opinion—that today TCD is the most promising non-invasive technique for continuous or intermittent quantitative ICP evaluation, especially in the emergency care or Neuro-ICU settings. It is clear the quest for “fine tuning” of this TCD application is still not over but even today quantitative and qualitative changes in TCD measured CBFV’s and PI’s values and TCD waveform morphologies may persuade physicians to undertake other diagnostic steps or to change medical treatment, thereby improving care of patients and their outcomes. To conclude, TCD can be used by experienced examiners to evaluate the presence of high ICP/low CPP and TCD as a non-invasive and simple procedure could be engaged in the daily management of TBI patients when ICH is suspected and/or must be confirmed; although more studies are needed before we can substitute direct invasive measurement of the ICP with TCD.
TCD AND CEREBROVASCULAR REACTIVITY
The brain’s response to the long-term effect of mTBI is only partially understood. It is well known and described that TBI has been inducing cerebral vascular dysfunction that is reflected in altered responses to various vasodilators. Cerebrovascular reactivity (CVR) is a key factor in regulating blood flow into the brain, and also represent a marker for the status of autonomic nervous system. If the brain’s regulatory system is not working, testing of CVR is one method of assessing the brain’s regulatory capabilities. TCD modality is a very convenient and non-expensive tool to measure CVR. In this method, carbon dioxide in the blood is transiently increased (such as with the holding of breath), and the breath-holding index (BHI) can be easily calculated. The breath-holding maneuver is a useful and well-tolerated screening method for CVR and was validated in numerous per reviewed publications.125-130 While numerous studies have focused on the acute period of brain parenchymal, behavioral, and vascular changes associated with mTBI, few have followed these changes over a more prolonged, chronic course of injury, or have attempted to correlate these changes with any enduring morbidity, like post-traumatic stress disorder. On the vascular front, there were significant contributions to the understanding of the brain’s microvascular response to injury, illustrating that in the early hours post-injury the cerebral microcirculation shows impaired vascular reactivity to known vasodilator challenges.49,130 Further, research suggested that this impaired CVR may persist for at least 1 week post-injury;130-133 however, beyond this period nobody followed the persistence of these vascular abnormalities. While previous reports have focused primarily on the short-term vascular alterations, nobody yet attempted to correlate these alterations with any persisting behavioral changes or potential therapeutic modulation. Reproducibility and non-invasive testing capabilities of TCD supports studies for its use as a diagnostic tool for mild TBI and marker for recovery.
CONCLUSIONS
The high sensitivity of TCD to identify abnormally high CBFV’s and PI’s due to the onset on of PTV and ICH, respectively, demonstrates that TCD is an excellent first-line examination to determine those patients who may need urgent aggressive treatment and potentially continuous invasive ICP monitoring. Because PTV and ICH represent significant events in a high proportion of patients after moderate and severe TBI, daily TCD monitoring is recommended for the management of such patients. In the contemporary neurointensive care of patients with TBI where cerebral hemodynamics can be disturbed or impaired basic neurological monitoring should include TCD. Growing evidence clearly supports the integration of daily TCD examinations to unmask otherwise occult alterations and to differentially adapt the type, extent, and duration of therapeutic interventions. By expanding our knowledge and experience, the integration of TCD in daily clinical routine will provide us with the means to improve outcome, which has not been possible by relying only on neurological examination alone as practiced in the past. Therefore, there is a potential for much broader utilization of TCD for patients after mild, moderate and severe TBI. To conclude, TCD ultrasonography represent a reliable, quantitative and non-expensive “biomarker” to the clinical manifestations of moderate and severe TBI and evaluation of patients with mTBI.
Notes
Ethics Statement
The Institutional Review Board process and patient consents were not proceeded because of a review article.
Availability of Data and Material
The data that support the findings of this study are available in the text.
Sources of Funding
None.
Conflicts of Interest
No potential conflicts of interest relevant to this article was reported.
Acknowledgements
None.

