Optic neuritis is defined as inflammation that damages the optic nerve. Common symptoms include eye pain and temporary vision loss. Optic neuritis is often idiopathic and can occur in association with demyelinating diseases, other rare autoimmune diseases, infectious diseases, and inflammatory and immune responses to vaccination.1 In adults, optic neuritis is mainly associated with autoimmune diseases such as multiple sclerosis and typically occurs in one eye. In contrast, post-infectious optic neuritis is common in children and mainly involves both eyes.2
Bilateral optic neuritis associated with central nervous system (CNS) infection is rare in adults. Only a few cases of optic neuritis post-encephalitis have been reported in the recent years.3,4 Cases of optic neuritis accompanying encephalitis and meningitis are even more rare. We would like to present a case report of bilateral optic neuritis, which is suspected to be related to CNS infection in an adult.
CASE REPORT
A 62-year-old woman was admitted with sudden bilateral visual loss since 3 days. She had also had headache, dizziness and fever for 4 days. She was receiving medication for hypertension and had no history of going abroad.
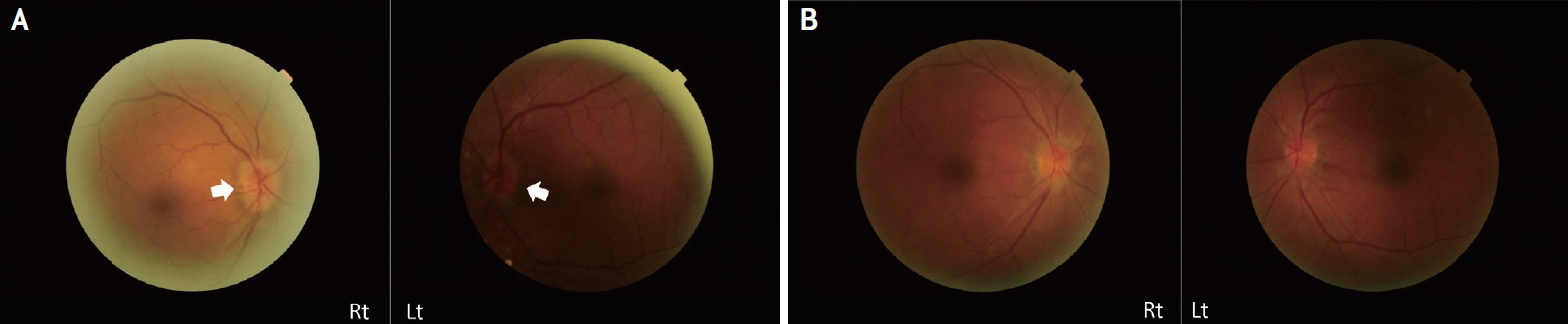
On physical examination, she had a mild fever of 37.8°C. Neurological examination showed no specific findings involving the motor and sensory systems. Meningeal irritation sign was negative, and all functions of cranial nerves were normal. On ophthalmologic examination, she retained some light perception on bilateral visual acuities. The pupils were equal, round, and reactive to light. A relative afferent pupillary defect was not definite in both eyes. Furthermore, there was no limitation or pain on eye movement. Dilated fundus evaluation revealed the presence of blurred disc margins, which led to a suspicion of papilledema (Fig. 1A).
Routine blood chemistry showed normal results. Additional laboratory tests including those for fluorescent antinuclear antibody (FANA), p-anti-neutrophil cytoplasmic antibody (ANCA), c-ANCA, anti-cardiolipin antibody, anti-beta-2 glucose 6 phosphate isomerase (GPI) antibody, vitamin B1, B6, B12, folate, anti-aquaporin 4 antibody, and paraneoplastic autoantibody were also showed normal results. Tests involving N-methyl-D-aspartate receptor (NMDAR), voltage gated potassium channels, and α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) were not performed because of the lack of encephalitis characteristics accompanying the optic neuritis. In contrast, thyroid function tests showed a decreased level of thyroid stimulating hormone of 0.223 uIU/mL, increased anti-microsome antibody level of more than 1,300 U/mL, and increased anti-thyroglobulin antibody level of 83.8 U/mL. However, triiodothyronine (T3) and free thyroxine (T4) levels were within the normal range.
The results showed delayed P100 latencies in full field visual evoked potential (VEP) bilaterally. These findings were indicative of prechiasmatic conduction defect in bilateral VEP pathways. Cerebrospinal fluid (CSF) examination was performed: CSF opening pressure, 15 cmH2O; white blood cell (WBC) count, 140/mm3 (79% lymphocytes); protein, 175.0 mg/dL; and glucose, 64 mg/dL (serum glucose: 115 mg/dL). There was no viable cell on CSF cytology. No oligoclonal band was detected in the CSF. Brain magnetic resonance imaging (MRI) showed diffuse swelling and heterogeneous enhancement in both optic nerves, which suggested infectious optic neuropathy (Fig. 2A). In addition, diffuse leptomeningeal enhancement and multiple hyperintensity lesions indicated meningitis and encephalitis (Fig. 2B). However, none were confirmed in the viral polymerase chain reaction (PCR), including herpes simplex virus types 1 and 2, Epstein-Barr virus, Enterovirus, cytomegalovirus culture, and varicella zoster virus culture. In addition, antibody-based screening test for tuberculosis, parasites, fugal, and bacterial infections, including Japanese encephalitis and Lyme disease, showed negative results.
She was treated with intravenous methylprednisolone and acyclovir for infectious optic neuritis and meningoencephalitis. After 9 days of treatment, brain MRI showed improved meningoencephalitis and optic neuritis (Fig. 2C, D). Follow-up CSF findings were as follows: opening pressure 15 cmH2O, WBC 100/mm3 (95% lymphocytes), protein 109.0 mg/dL, and glucose 77 mg/dL (serum glucose 248 mg/dL). CSF cytology showed few macrophages. Two weeks after treatment, her vision improved, and she was able to read text; the bilateral papilledema also improved (Fig. 1B). Two months later, her vision was intact, and the disc swelling in both eyes also improved.
DISCUSSION
There are various possible causes of spontaneous bilateral optic neuritis. First, optic neuritis is often a symptom of demyelinating diseases such as multiple sclerosis or neuromyelitis optica spectrum disorders (NMOSD).2 In this case, brain MRI findings were consistent with meningoencephalitis. No spinal cord abnormalities were found on spine MRI. In addition, tests for the oligoclonal band of CSF and anti-aquaporin 4 antibody were performed twice to exclude multiple sclerosis and NMOSD but showed negative results. Second, the possibility of other autoimmune diseases should be considered. Thyroid function tests revealed subclinical hyperthyroidism,5 including elevated anti-microsome antibody and anti-thyroglobulin levels. When optic neuropathy is associated with thyroid dysfunction, it is called dysthyroid optic neuropathy (DON). DON is mainly related to Graves’ disease, and its mechanism is explained by compression of the optic nerve caused by extraocular muscle enlargement.6 In this case, it is difficult to consider DON because the swelling of the medial rectus muscle was not confirmed in the brain MRI (Fig. 2A). In addition, although there are studies showing the relationship between antithyroid antibodies and demyelinating diseases,7 we have previously explained that this case is less likely to be associated with any demyelinating disease. Third, optic neuritis due to infectious disease or inflammatory reaction should be considered. Optic disc swelling on fundoscopy and the swelling and enhancement of the optic nerve on brain MRI suggest papillitis, which can be caused by almost all infections.8 Although there is no confirmed infectious source, it is most likely that the diagnosis is infectious optic neuritis accompanying meningoencephalitis, when considering the imaging test results, CSF findings, and clinical symptoms.
Since it is clinically difficult to identify the cause of atypical optic neuritis, clinicians should try to perform active screening and appropriate treatment. Intravenous corticosteroids have been effective in most optic neuritis patients.1-4,8 When infectious optic neuropathy is suspected, as in this case, it is helpful to use antiviral drugs and steroids with caution.8 In conclusion, it is very important for the clinician to perform the necessary tests and initiate appropriate treatment as soon as possible for patients with visual impairment of unknown cause.