Hypertensive encephalopathy is characterized by neurological symptoms including severe hypertension, nausea, vomiting, headache, visual abnormalities, mental changes, and seizures. Hypertensive encephalopathy is usually treated with blood pressure adjustment. The classic magnetic resonance imaging (MRI) markers of hypertensive encephalopathy are symmetric high signal intensities in the subcortical parieto-temporo-occipital regions on T2-weighted imaging (T2WI) and fluid-attenuated inversion recovery (FLAIR) imaging. Approximately, two-thirds of patients also have high signal intensity in the frontotemporal lobes, referred to as typical hypertensive encephalopathy. High signal intensity is rarely seen in the brainstem, cerebellum, or basal ganglia, and when seen it is referred to as atypical hypertensive encephalopathy.1
In hypertensive encephalopathy, the intensity in the parieto-occipital lobe is the most common lesion, most commonly seen in the frontal lobe, followed by the temporal lobe, cerebellum, brainstem, and basal ganglia. Both the brainstem and basal ganglia are rarely affected. Here, we report the case of a woman diagnosed with hypertensive encephalopathy involving the brainstem, basal ganglia, and cerebral white matter.
CASE
A 51-year-old woman with no significant past medical history presented to the emergency department with a one-day history of left limb weakness, dysarthria, and headache. She denied any recent fever, upper respiratory tract infections, and had not received any recent vaccinations. Her only significant medical history included intensive care unit hospitalization in her mid 20s secondary to a motor vehicle accident and two spontaneous abortions in her early 40s. She was a social drinker and a non-smoker. Three days before presentation, she had a headache with associated nausea and vomiting. She visited the emergency department one day before hospitalization and while there her temperature was 36.0℃, blood pressure was 140/90 mmHg, heart rate was 75 BPM, and her respiratory rate was 20 breaths/min.
Neurological examination was significant for mild dysarthria and left upper/lower extremity motor weakness with motor grade 4. Babinski sign was positive in her left lower extremity. The rest of her neurological examination was normal. The following day the patient presented to the outpatient clinic with a blood pressure of 195/107 mmHg. The rest of her examination was normal. To rule out hemorrhagic stroke, a computed tomography scan was performed immediately after admission and it showed small vessel disease in the basal ganglia and thalamus, as well as changes in her periventricular white matter.
Serum electrolytes, glucose, C-reactive protein, free T4, and thyroid-stimulating hormone were normal, as well as liver and renal function tests, complete blood counts with differentials, and erythrocyte sedimentation rate. Her visual acuity and visual fields were unchanged although she complained of pain in her eyeball. Funduscopic exam (Fig. 1) was performed to check for ocular changes, however, no specific funduscopic findings, except for a few dot hemorrhages, were noted in the bilateral fundi. Cerebrospinal fluid analysis was unremarkable for the presence of white or red blood cells. Notch3 gene mutation was not detected on genetic analysis.
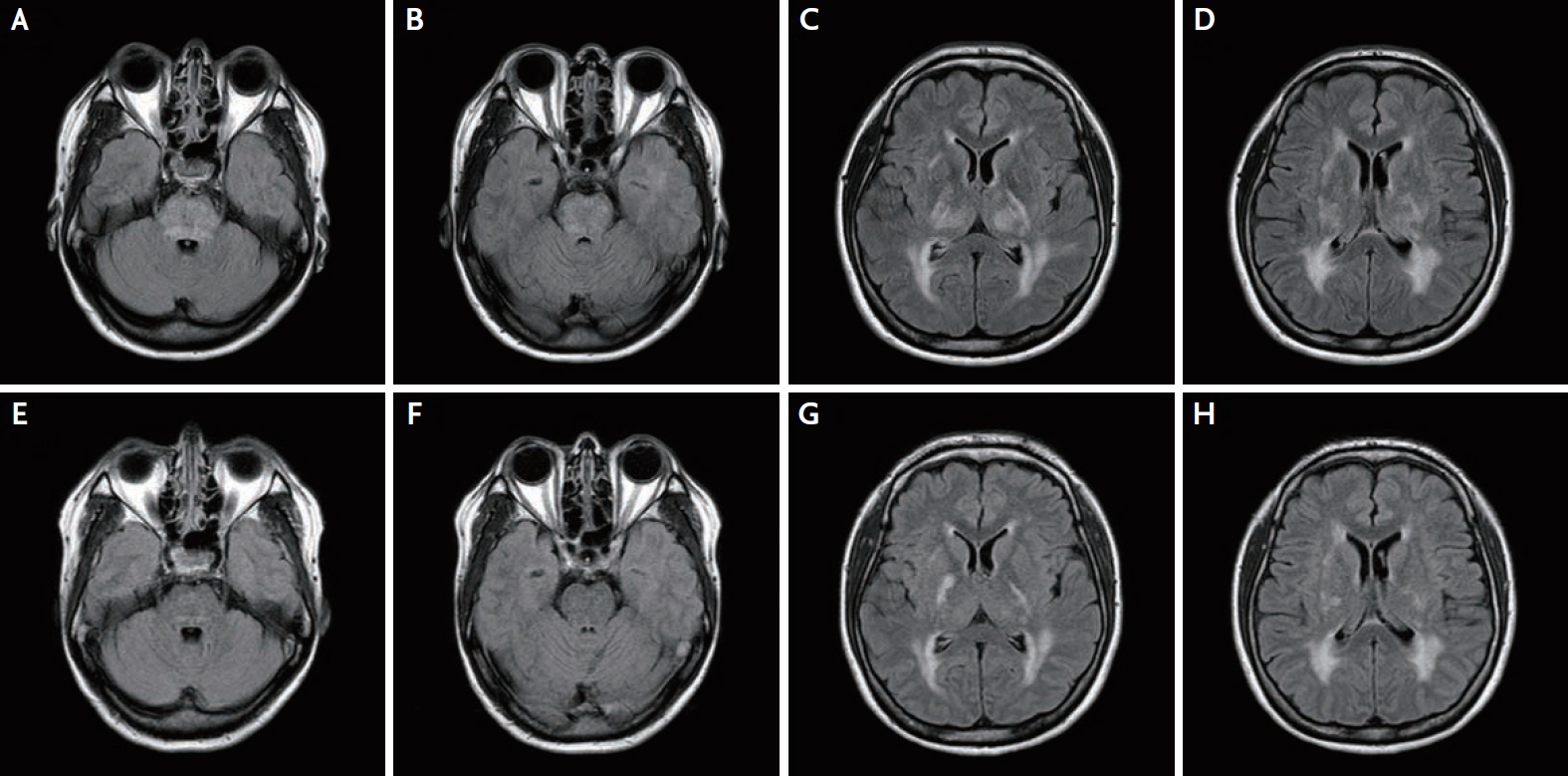
T2WI and FLAIR MRI revealed high signal intensities in the pons and basal ganglia, and in the parieto-occipital lesions (Fig. 2A-D). Diffusion MRI revealed that there was a small, restricted lesion in the right basal ganglia. These clinical features led to the correct diagnosis of hypertensive encephalopathy. After the patient’s blood pressure was successively managed with intravenous nicardipine and other oral medications, her symptoms, except for mild weakness in her left arm, resolved. During a follow-up MRI 6 days after her admission, the signal intensity in the pons and thalamus had normalized, except for that in the internal capsule and the parieto-occipital (Fig. 2E-H). The patient was then discharged from the hospital with a new oral hypertension medication.
DISCUSSION
Hypertensive encephalopathy classically involves the parieto-occipital white matter, and only rarely involves the brain stem. The pathophysiology of hypertensive encephalopathy is associated with vasogenic edema without evidence of cytotoxic edema or infarction. The blood vessels of the posterior circulation are innervated by sympathetic nerves. Therefore, in response to increased arterial pressure, vasoconstriction does not begin properly.2 Deep brain regions such as the basal ganglia, thalamus, and brain stem receive circulation from the branches of the middle cerebral artery or basilar artery. The cortex and subcortex are supplied from the terminal branch of these arteries, and thus experience less intense arterial blood pressure than deep structures.3 Thus, severe and sustained arterial blood pressure elevation can involve deep brain structures.
It is relatively easy to recognize typical hypertensive encephalopathy cases, while atypical cases are more difficult based on their imaging features alone. For example, osmotic demyelination syndrome can be confused with posterior reversible encephalopathy syndrome, especially if the brainstem is involved in hypertensive encephalopathy.4 Recognizing typical serum electrolyte abnormalities and causes such as rapid correction of hyponatremia can aid differentiation. Unlike hypertensive encephalopathy, central or extrapontine myelinolysis is often enhanced in the subacute phase of follow-up postcontrast MRI.5
Cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL) is an inherited microangiopathy characterized by recurrent ischemic episodes and cognitive deficits and is caused by mutations in the Notch3 gene located on chromosome 19.5 CADASIL is characterized by low signal intensity on T1-weighted MRI images and high signal intensity on T2-weighted images, usually with multiple confluent white matter lesions of various sizes. These lesions are concentrated around the basal ganglia, pons, and periventricular white matter. CADASIL is similar to hypertensive encephalopathy based on radiological features alone.6 A variety of other diseases, such as infarction, rhombencephalitis, and brain neoplasm, appears similar to hypertensive encephalopathy based on imaging features.7
Hypertensive encephalopathy is a reversible and treatable disease. A reduction of lesions during follow up MRI is an important differentiating marker of this condition, so it is crucial to include hypertensive encephalopathy in the list of differential diagnoses and perform the appropriate work-up and treatments.