A sudden onset neurological deficit is most likely due to an acute stroke. However, stroke mimics account for between a fifth and a quarter of suspected strokes.1 Previous studies have identified seizures, migraine, toxic/ metabolic diseases, and functional or conversion disorders as non-vascular diseases that can mimic stroke.1,2 Furthermore, several cases were reported on central nervous system (CNS) infections, such as meningitis, encephalitis, and cerebritis, manifesting as acute stroke-like symptoms. Since CNS infections frequently result in devastating consequences, the early diagnosis and treatment requires vigilance based on the history and physical examination which must be confirmed with appropriate neuroimaging and laboratory evaluation. Herein, we report a rare case of Klebsiella pneumoniae cerebritis with hyperacute middle cerebral artery (MCA) stroke-like symptoms in a patient with K. pneumoniae liver abscess syndrome (KLAS).
CASE
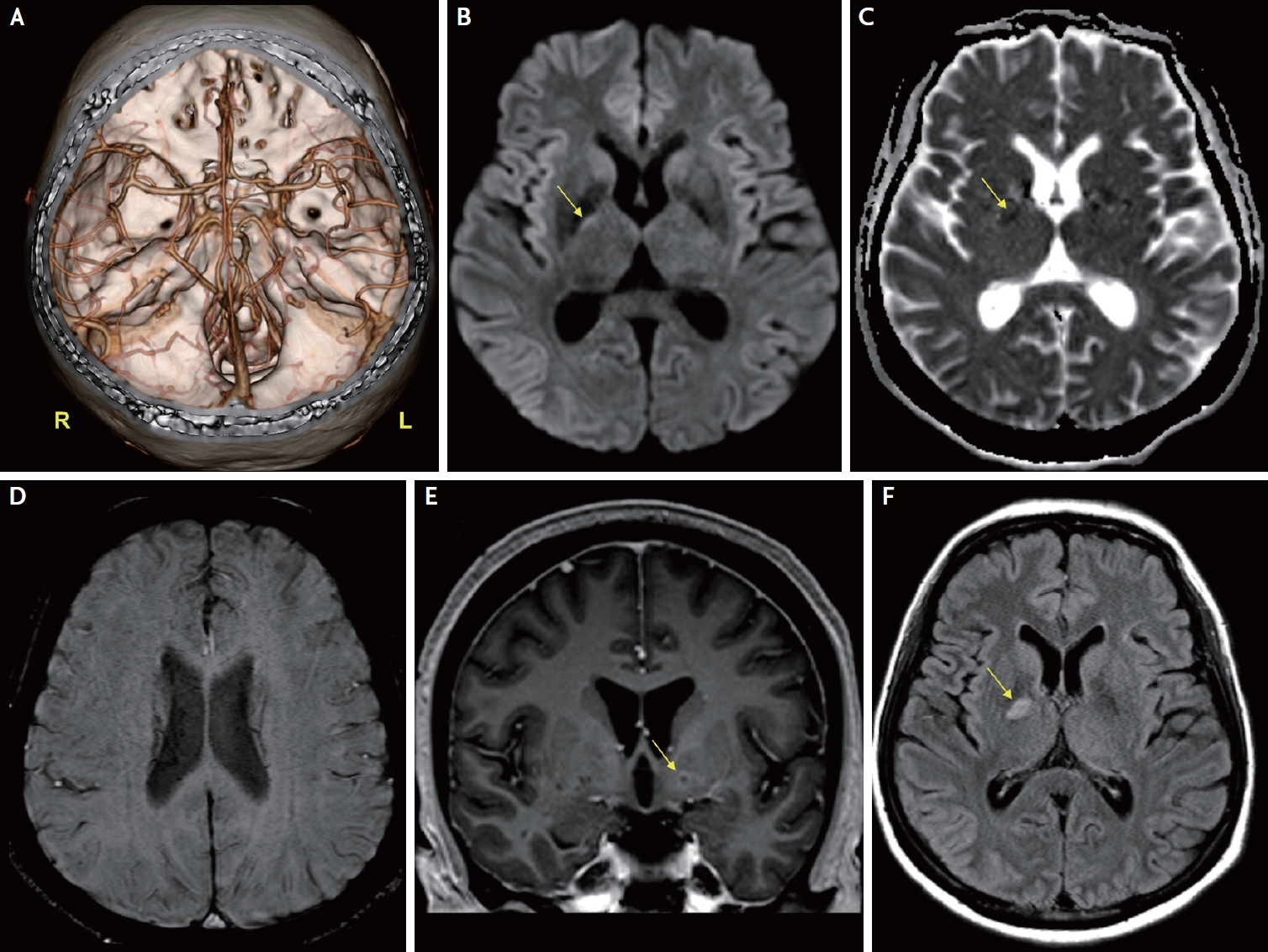
A 52-year-old woman was brought to the emergency department with complaints of sudden onset of left-sided weakness and confusion 2hours prior to her arrival. She had a history of diabetes, but was not under any medications. Her neurological examination revealed a profound left-sided neglect and conjugate deviation of the eyes to the right side. A motor examination revealed Medical Research Council (MRC) grade 2 power in the left upper and lower extremities, and the Babinski sign was positive on the left side. Her National Institutes of Health Stroke Scale score was 8. Despite stable vital signs and no fever (36.7°C), the patient had nuchal rigidity. Acute MCA stroke was suspected preferentially, and a neuroimaging diagnostic workup was immediately conducted. Initial brain computed tomography (CT) showed no ischemic or hemorrhagic lesion, and CT angiography found no steno-occlusive lesions (Fig. 1A). Further, perfusion CT revealed no perfusion defects in the brain. However, a brain magnetic resonance imaging (MRI) demonstrated a focal hyperintense lesion in the posterior limb of the right internal capsule on diffusion-weighted imaging (Fig. 1B). This lesion was confirmed to be an acute ischemic infarction based on the low signal intensity on the apparent diffusion coefficient map (Fig. 1C). There were no abnormal findings on the susceptibility-weighted imaging (Fig. 1D).
Considering the discrepancy between the clinical symptoms, which suggested a right MCA stroke, and the neuroimaging findings, we terminated further hyperacute thrombolytic therapy and performed an extensive laboratory diagnostic workup. The results of the laboratory tests were as follows: white blood cells, 16,960/μL (4,000–10,000/μL, neutrophils, 85.8%; lymphocytes, 5.4%; and monocytes, 4.2%); aspartate aminotransferase, 129 IU/L (<35 IU/L); alanine aminotransferase, 111 IU/L (<35 IU/L); alkaline phosphatase, 204 IU/L (30–120 IU/L); glucose, 548 mg/dL (74–106 mg/dL); hemoglobin A1c, 11.6%; triglyceride, 544 mg/dL (<150 mg/ dL); erythrocyte sedimentation rate, 119 mm/h (0–20 mm/h); and C-reactive protein, 35.6 mg/dL (<0.5 mg/dL). A cerebrospinal fluid (CSF) analysis revealed the following: a pressure of 150 mmH2O; turbid color; 7,000 white blood cells (neutrophils, 67%; monocytes, 26%; and lymphocytes, 7%); protein, 276 mg/dL (15–130 mg/dL); glucose, 213 mg/dL; and chloride, 119.9 mmol/L. An abdominal enhanced CT demonstrated an irregularly shaped, heterogeneously low attenuated lesion at S7 in the liver (approximately 4.5×8.2×6.4 cm), which was consistent with a radiologic finding of liver abscess. Based on these findings and a history of uncontrolled diabetes in the patient, we suspected KLAS and initiated empirical antibiotic treatment with 2 g of ceftriaxone every 12 hours and 500 mg of metronidazole every 8 hours. In addition, she was administered 100 mg of aspirin for secondary stroke prevention.
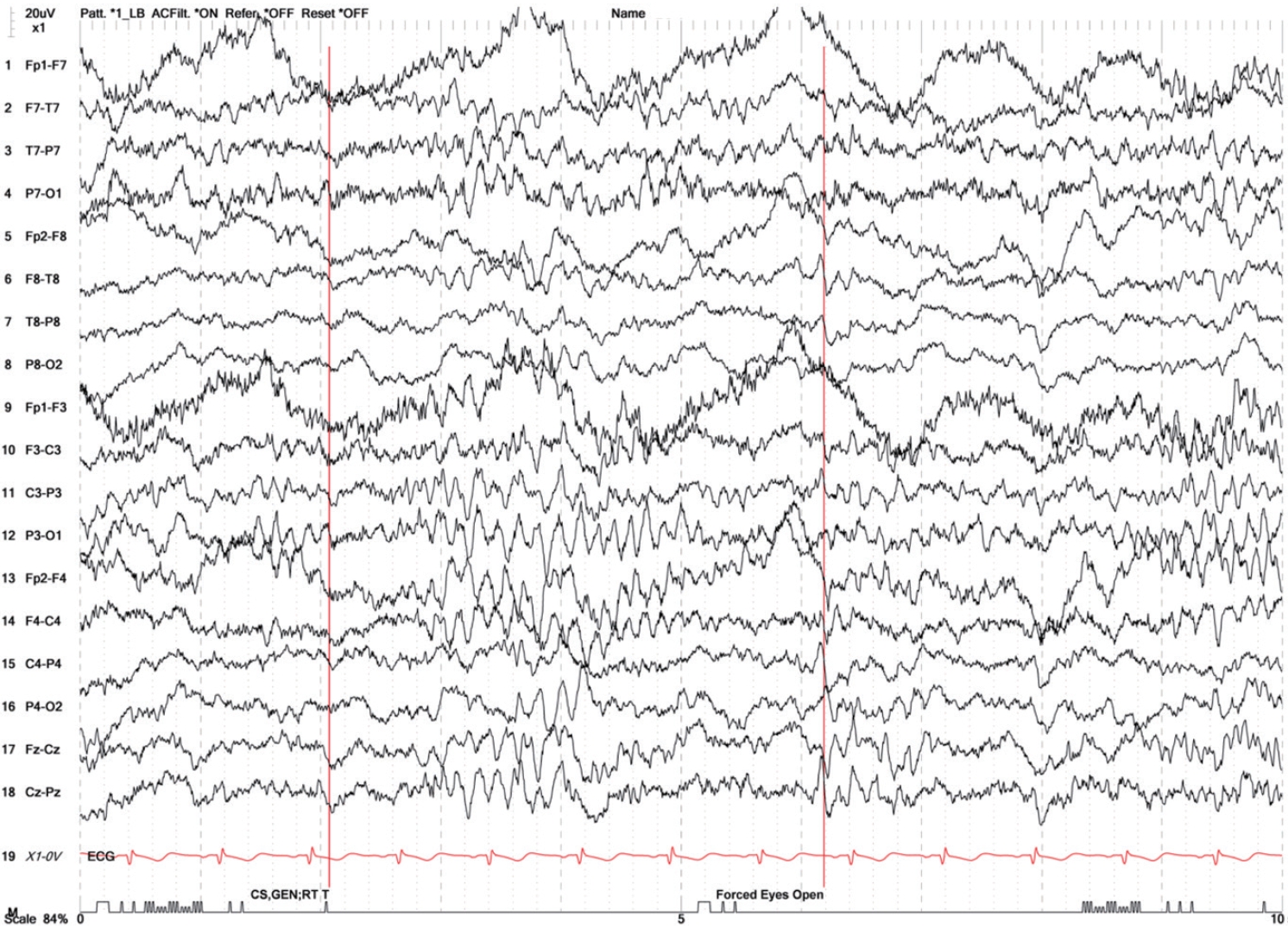
On the second day of hospitalization, a mass spectroscopy-based direct identification of bacteria in the blood and urine cultures revealed K. pneumoniae. The patient was alert, and all neurological deficits suggesting right MCA stroke was improved (approximately 5 hours after symptom onset), except for mild left-sided hemiparesis (MRC grade 4+). Interventional radiology-guided liver abscess drainage with percutaneous drain placement was performed while continuing the antibiotic treatment. On the fourth day of hospitalization, K. pneumoniae was confirmed in the blood, sputum, and CSF cultures. Further examination provided no evidence of lung or ocular involvement of the disease. However, the patient’s brain MRI revealed a subtle diffuse leptomeningeal enhancement and a suspicious small rim-enhancing lesion in the left basal ganglia on contrast-enhanced T1-weighted imaging (Fig. 1E). In addition, a hyperintense lesion in the posterior limb of the right internal capsule was found on the fluid-attenuated inversion recovery sequence, confirming the acute cerebral infarction observed on the initial diffusion-weighted imaging (Fig. 1F). Electroencephalography (EEG) indicated a generalized continuous slowing with continuous regional slowing from the right temporal lobe (Fig. 2). Based on these clinical and neuroimaging findings, the patient was diagnosed with cerebritis associated with KLAS. After one month of hospitalization, the patient fully recovered from KLAS and was discharged from our hospital without any neurological deficits.
DISCUSSION
K. pneumoniae is a gram-negative, capsulated, non-motile, gas-producing enteric bacillus present in the normal flora of the human intestine and oral cavity. KLAS is clinically characterized by bacteremia, liver abscesses, and metastatic infections caused by hypervirulent strains of the capsular K1 or K2 serotype.3 Fever, chills, and abdominal pain are the most common clinical symptoms among patients with KLAS; however, they are not characteristic of invasive K. pneumoniae infection.3 Metastatic KLAS infection commonly affects the lungs, CNS, and eyes.3,4 Diabetes is not only a well-known concomitant disease in patients with KLAS,5 but also an independent risk factor for metastatic infection.4
To the best of our knowledge, this is the first report of K. pneumoniae cerebritis presenting with symptoms resembling a hyperacute MCA stroke in a patient with KLAS. The patient’s localized neurological deficits, including left-sided weakness, neglect, and conjugate deviation of the eyes to the right side, which had occurred only 2 hours earlier, suggested a hyperacute right MCA stroke. We could immediately perform multimodal neuroimaging on the patient, which enabled us to recognize the discrepancy between the neurological deficits and neuroimaging findings and diagnose K. pneumoniae cerebritis at an early stage. Considering that severe CNS infection could present as acute stroke6 and the high mortality of metastatic CNS infection in patients with KLAS7 a multimodal neuroimaging approach that could be rapidly performed in a short time in clinical practice is critical for discriminating acute stroke mimics.
Previous studies have suggested that paroxysmal septic emboli, vasospasm, and vasculitis are the pathophysiological mechanisms causing a stroke-like presentation in bacterial CNS infection8,9 It was interesting that a lateralized and localized MCA syndrome, which was supported by EEG findings, occurred prominently at the early stage of K. pneumoniae cerebritis. However, there was no asymmetric perfusion defect on the initial perfusion CT; this necessitates further investigation with more such cases to elucidate the mechanism that could explain rare symptoms in this patient. Appropriate antibiotic treatment is the key to survival in patients with bacterial CNS infection10 Thus, clinicians should consider K. pneumoniae cerebritis as a differential diagnosis upon detecting a liver abscess in patients with uncontrolled diabetes and acute neurological deficits.