 |
 |
- Search
| J Neurosonol Neuroimag > Volume 15(2); 2023 > Article |
|
Abstract
Thrombus resolution is generally associated with a favorable prognosis. Patients with thrombus resolution often present with erythrocyte-rich thrombi, frequently associated with large artery atherosclerosis or alteplase use. This might explain the increased rate of cervical carotid artery occlusion and reduced incidence of atrial fibrillation in patients with thrombus resolution. However, recent reports have demonstrated that thrombus resolution can occur owing to several etiologies and various factors. We present two cases of thrombus resolution: one associated with internal carotid artery dissection and the other with a left atrial appendage thrombus. Notably, both were considered to be unrelated to large-artery atherosclerosis or alteplase use.
Changes in the thrombus location are common in patients with acute ischemic stroke. Complete thrombus resolution is rare but desirable, as it usually has a favorable prognosis.1 Thrombus resolution is reported to occur frequently after the intravenous administration of alteplase.2,3 Recent reports have demonstrated that thrombus resolution can occur due to several etiologies and various factors.4-7 We report two patients with acute ischemic stroke who experienced complete thrombus resolution, which may not be related to large-artery atherosclerosis or alteplase use.
A 29-year-old female was admitted to our hospital with complaints of left-sided weakness (Medical Research Council scale of upper and lower limbs, 4+/4+), facial palsy, and flaccid dysarthria that occurred 1 hour ago. Both her personal medical history and that of the family were unremarkable, and no history of head trauma was present. Her electrocardiogram (EKG) revealed normal sinus rhythm, and the vital signs too were stable, with a blood pressure of 127/78 mmHg. A neurologic examination revealed mild left arm weakness and left central facial palsy. The initial National Institutes of Health Stroke Scale (NIHSS) score was 3.
Magnetic resonance imaging performed 2 hours after onset of symptoms revealed an acute lesion in the right middle cerebral artery (MCA) (Fig. 1A). T2 fluid-attenuated inversion recovery (FLAIR) images indicated a high-signal-intensity lesion in the right Sylvian fissure, suggesting an intra-arterial thrombus (Fig. 1B). Susceptibility-weighted angiography also displayed hypointense signals with blooming, indicating thrombus formation (Fig. 1C). Computed tomography angiography (CTA) of the head and neck revealed right MCA M2 (superior division) occlusion and right proximal internal carotid artery (ICA) dissection (Fig. 1D). Moreover, CTA axial images also displayed a double-lumen sign and an intimal flap (Fig. 1E). Thrombolysis was not performed as the patient’s symptoms were mild and rapidly resolved. Dual-antiplatelet therapy (DAPT) and 40 mg atorvastatin were prescribed after a loading dose of 300 mg aspirin and 300 mg clopidogrel, respectively.
Laboratory test results for hypercoagulable causes were negative. Other serological parameters, including erythrocyte sedimentation rate and C-reactive protein levels, were within normal limits. Additionally, a standard 24-hour Holter electrocardiogram and transthoracic echocardiogram were also within normal limits.
The dysarthria and facial palsy gradually improved after admission. Four days after symptom onset, repeat T2 FLAIR and CTA demonstrated recanalization of the previously occluded right MCA M2 (superior division), although focal stenosis/luminal irregularity in the right MCA M2 persisted (Fig. 1F).
At discharge, we prescribed a maintenance regimen consisting of DAPT and atorvastatin. One month after symptom onset, her condition remained stable with only mild residual dysarthria.
A 50-year-old male was referred to our center with a 1-hour history of dizziness and mental status changes. His medical history was significant for sick sinus syndrome 2 years prior, for which he had undergone implantable cardioverter-defibrillator (ICD) insertion. He was prescribed edoxaban 60 mg, however, direct oral anticoagulants (DOAC) were withheld for 5 days due to ICD insertion. On examination, the patient appeared confused and drowsy. Moreover, the EKG revealed a paced ventricular rhythm. His vital signs were stable with a blood pressure of 153/72 mmHg. A neurological examination revealed left-sided weakness (grade IV), right-sided ataxia, and dysarthria. The initial NIHSS score was 8. Furthermore, gaze-evoked nystagmus was also identified during extraocular movement testing.
Additionally, CTA demonstrated an 8 mm-long intraluminal thrombus in the distal portion of the right vertebral artery (VA) (posterior inferior cerebellar artery origin) (Fig. 2A, B). Therefore, the patient underwent thrombolysis. Subsequently, transfemoral cerebral angiography (TFCA) revealed a recanalized right distal VA without basilar or posterior cerebral artery occlusion (Fig. 2C, D). The patient was restarted on DOAC.
His upper and lower extremity motor weakness resolved on day 2 after admission. No abnormal extra-ocular movements were observed. His pupils were symmetrical, palatal function was normal, and no ptosis or hoarseness was noted. Echocardiography displayed an ejection fraction of 53% and an echogenic mass (2.84 × 1.43 cm) that almost filled the left atrial (LA) appendage. The mass was located approximately 0.4 cm from the LA appendage inlet. We suspected an LA appendage thrombus and prescribed DOACs at discharge. At the time of his 2-month follow-up, his symptoms had not recurred. Three months after symptom onset, follow-up echocardiography revealed the resolution of the LA appendage thrombus.
Thrombi are classified as white or red based on their appearance and predominant color. White thrombi may be rich in platelets or fibrin, whereas red thrombi typically contain blood cells. We reasoned that fibrin- and erythrocyte-rich red thrombi were more likely to resolve than platelet-rich white thrombi.4,5 Spontaneous recanalization rates are greatly influenced by stroke etiology, with cardioembolism being the most frequent etiology in the recanalization group (44% vs. 25%), whereas large artery atherosclerosis was more common in the non-recanalization group (38% vs. 18%).7 Additionally, in patients with ICA occlusion due to arterial dissection, the recanalization process may occur between 2 days and 6 weeks after stroke.6 The arterial dissection provides opportunities for thrombus formation and ischemic strokes. They occur secondary to arterial dissection and are usually embolic; 92.2% of cerebral infarctions associated with ICA dissection have an embolic origin leading to focal infarctions, including cortical and subcortical infarcts, and only 7.7% observed as watershed infarctions associated with reduced hemodynamic flow.8,9 This might help explain the resolution of MCA thrombus in case 1. To prove our assertion clearly, measuring the Hounsfield unit of non-contrast CT would be helpful; however, a hyperdense MCA sign could not be identified in our case.
The resolution of the cardioembolic thrombi in case 2 appears distinct. A randomized study demonstrated similar efficacy and safety of anticoagulant, antiplatelet, and dual antiplatelet regimens for extracranial cervical artery dissection.10 Although, case 1’s thrombus resolved following short-term DAPT administration, case 2 experienced spontaneous resolution. Cardioembolic thrombi should be considered, especially in rapidly changing oscillating shear stress environments, such as the LA appendage. The activation of the coagulation cascade constitutes the central mechanism of thrombus formation.11
Furthermore, whether thrombi of cardiac origin or those occurring secondary to arterial dissection resolve more readily than those occurring secondary to atherosclerosis remains still unknown. We present two cases of resolved thrombi originating from a cardioembolic source or secondary to arterial dissection. These cases challenge our understanding of the resolution of thrombi caused by several etiological factors. These findings imply that selecting drugs based on the mechanism of thrombus formation may improve prognosis and increase the likelihood of thrombus resolution. Future studies must determine which drugs used following thrombus histology induce better thrombus resolution.
NOTES
Fig. 1.
MRI and CTA of the brain. (A) DWI displays an acute lesion in the right MCA territory. (B, C) T2 FLAIR and SWAN images display intra-arterial thrombus in the right Sylvian fissure (white arrows). (D, E) CTA exhibits right proximal ICA dissection (red arrows). (F) Follow-up T2 FLAIR demonstrates recanalization of the previously occluded right MCA M2 (red arrow). MRI, magnetic resonance imaging; CTA, computed tomography angiography; DWI, diffusion weighted imaging; FLAIR, fluid attenuated inversion recovery; SWAN, susceptibility-weighted angiography; ICA, internal carotid artery; MCA, middle cerebral artery.
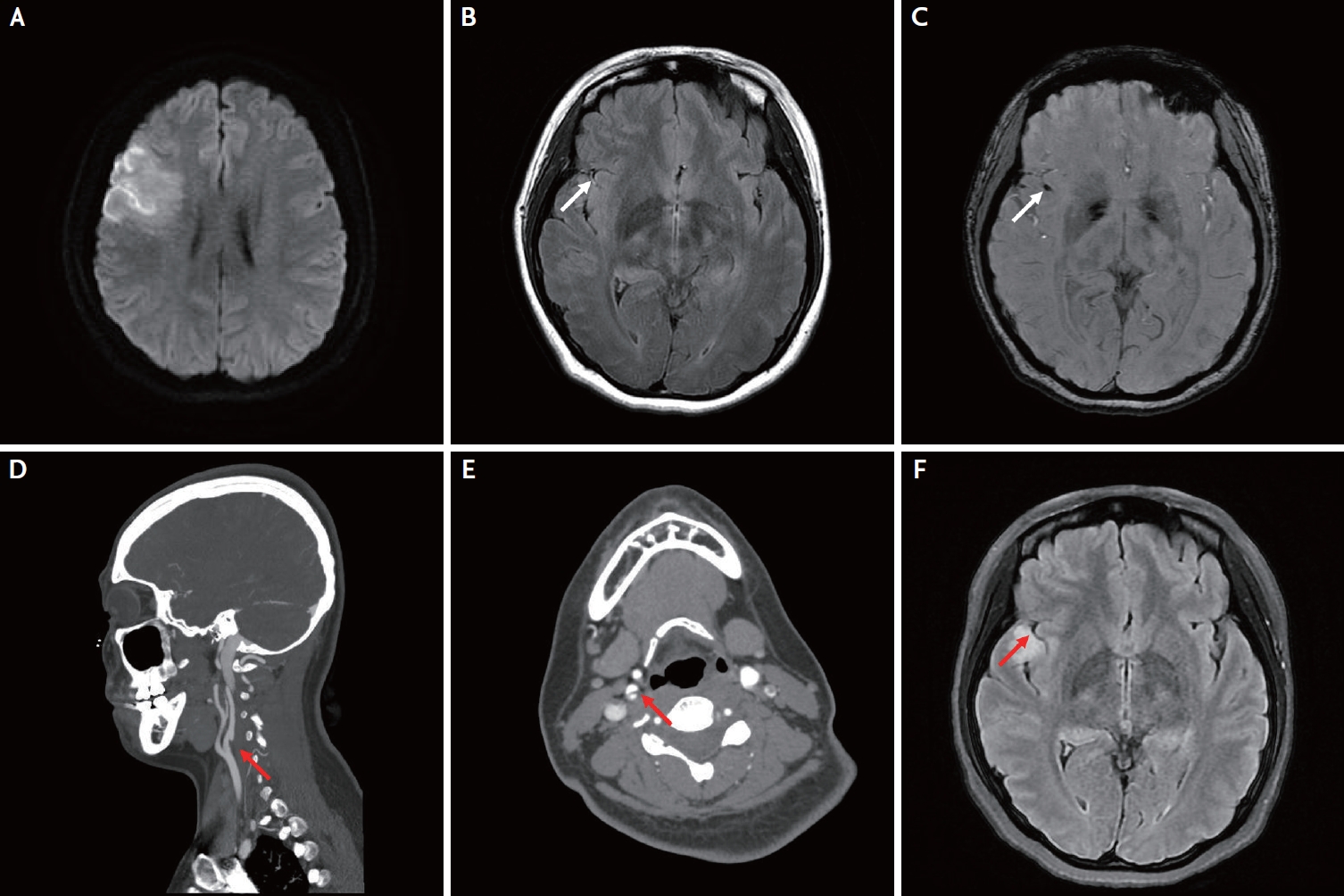
Fig. 2.
CTA and TFCA of head and neck. (A, B) CTA displays an 8 mm-long intraluminal thrombus in the right VA distal (PICA origin) portion (red arrows). (C, D) TFCA displays a recanalized right distal VA with no BA or PCA occlusion. CTA, computed tomography angiography; TFCA, transfemoral cerebral angiography; VA, vertebral artery; PICA, posterior inferior cerebellar artery; BA, basilar artery; PCA, posterior cerebral artery.

REFERENCES
1. Alves HC, Treurniet KM, Jansen IGH, Yoo AJ, Dutra BG, Zhang G, et al. Thrombus migration paradox in patients with acute ischemic stroke. Stroke. 2019;50:3156-3163.



2. Kim YD, Nam HS, Kim SH, Kim EY, Song D, Kwon I, et al. Time-dependent thrombus resolution after tissue-type plasminogen activator in patients with stroke and mice. Stroke. 2015;46:1877-1882.


3. Mueller L, Pult F, Meisterernst J, Heldner MR, Mono ML, Kurmann R, et al. Impact of intravenous thrombolysis on recanalization rates in patients with stroke treated with bridging therapy. Eur J Neurol. 2017;24:1016-1021.



4. Yuki I, Kan I, Vinters HV, Kim RH, Golshan A, Vinuela FA, et al. The impact of thromboemboli histology on the performance of a mechanical thrombectomy device. AJNR Am J Neuroradiol. 2012;33:643-648.



5. Niesten JM, van der Schaaf IC, van Dam L, Vink A, Vos JA, Schonewille WJ, et al. Histopathologic composition of cerebral thrombi of acute stroke patients is correlated with stroke subtype and thrombus attenuation. PLoS One. 2014;9:e88882.



6. Caso V, Paciaroni M, Corea F, Hamam M, Milia P, Pelliccioli GP, et al. Recanalization of cervical artery dissection: influencing factors and role in neurological outcome. Cerebrovasc Dis. 2004;17:93-97.



7. Vanacker P, Lambrou D, Eskandari A, Ntaios G, Cras P, Maeder P, et al. Improving the prediction of spontaneous and post-thrombolytic recanalization in ischemic stroke patients. J Stroke Cerebrovasc Dis. 2015;24:1781-1786.


8. Kim YK, Schulman S. Cervical artery dissection: pathology, epidemiology and management. Thromb Res. 2009;123:810-821.


9. Lucas C, Moulin T, Deplanque D, Tatu L, Chavot D. Stroke patterns of internal carotid artery dissection in 40 patients. Stroke. 1998;29:2646-2648.


10. Markus HS, Levi C, King A, Madigan J, Norris J; Cervical Artery Dissection in Stroke Study (CADISS) Investigators. Antiplatelet therapy vs anticoagulation therapy in cervical artery dissection: The Cervical Artery Dissection in Stroke Study (CADISS) randomized clinical trial final results. JAMA Neurol. 2019;76:657-664.










